Struvite Control and Recovery
A Theoretical and Practical Evaluation
Last Modified Aug 02, 2022
The Miami-Dade Water and Sewer Department’s (MDWASD) operating staff has identified struvite deposits in the solids handling process equipment at the Central District Wastewater Treatment Plant (CDWWTP) and the South District Wastewater Treatment Plant (SDWWTP).
Struvite formation is a common problem in wastewater treatment plants and can cause significant damage to equipment, pumps, and piping. It is important to address any struvite problems as a significant struvite accumulation can lead to biosolids treatment mechanical and process failures. Struvite deposits have been identified in the digester units, the digester piping system including valves and heat exchangers and the centrifuge dewatering units downstream of the digester system at both plants.
Currently, MDWASD is feeding Ferric Sulfate (Fe2(SO4)3 to the influent of the centrifuges at the CDWWTP as a short term fix for resolving struvite precipitation issues as well as improving the solids capture rate. Ferric is used to bind up the soluble orthophosphate preventing it from forming struvite. This solution, however, has been costly with an estimated ferric sulfate cost of $50,000 per month. The SDWWTP is currently spending approximately $30,000 per month in maintenance labor directly related to struvite issues.
Related Solutions:
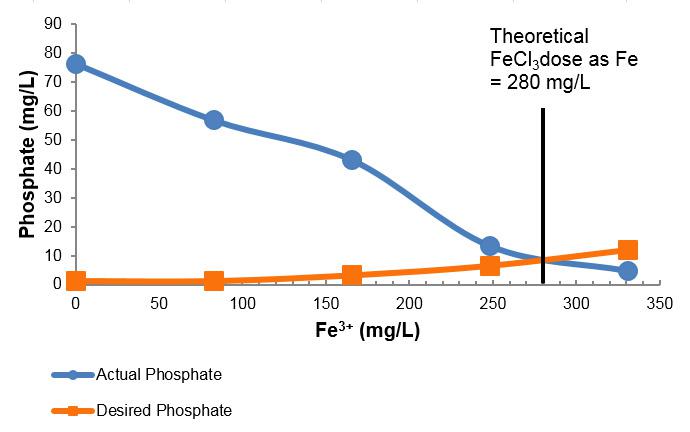
As a result, an extensive evaluation was conducted to develop several short and long term solutions to struvite precipitation, including chemical and sidestream treatment. Initially, a series of batch tests were carried out to determine the best type and dose of metal salt to use to control struvite formation in the digested sludge and centrate obtained from Miami Dade’s WWTPs. The batch test results indicated that for the SDWWTP centrate a Ferric Chloride dose of 285 mg/L as FE or a Ferric Sulfate dose of 275 mg/L as FE were optimal for controlling struvite formation. Similarly for the CDWWTP centrate, a Ferric Chloride dose of 280 mg/L as Fe and a Ferric Sulfate of 325 mg/L as Fe were found to be optimal.


In conjunction, ductile iron scaling coupons were placed (inside pipes) in various locations (primary and secondary digester effluent, feed to dewatering and centrate wet well) at both the CDWWTP and the SDWWTP to measure the degree of struvite formation and the effectiveness of applied doses of iron salts determined from the batch tests. These coupons were removed at 40 and 90 days and were visually inspected and analyzed using stereoscopic imaging.

As part of the analysis of long-term solutions to struvite precipitation, sidestream treatment of the centrate was evaluated, specifically the Ostara Nutrient Recovery Process (Pearl®) through pilot testing. The Pearl® process takes the nutrient rich recycle stream and adds magnesium chloride and caustic to intentionally precipitate struvite pellets that can then be sold as premium quality fertilizer. The pilot test results demonstrated that the Pearl® process is effective at removing orthophosphate with removal performance averaging 86% on the SDWWTP centrate and 76% on the CDWWTP centrate.
A BioWin process simulation model was also used to evaluate site specific struvite precipitation potential for the different long-term struvite reduction alternatives for the SDWWTP and CDWWTP, including Fe addition and sidestream treatment (Pearl®). The results of the model will be presented in the paper and oral presentation.
Finally, a 20-year net present worth cost evaluation was conducted for each alternative and the results were shared with MDWASD.